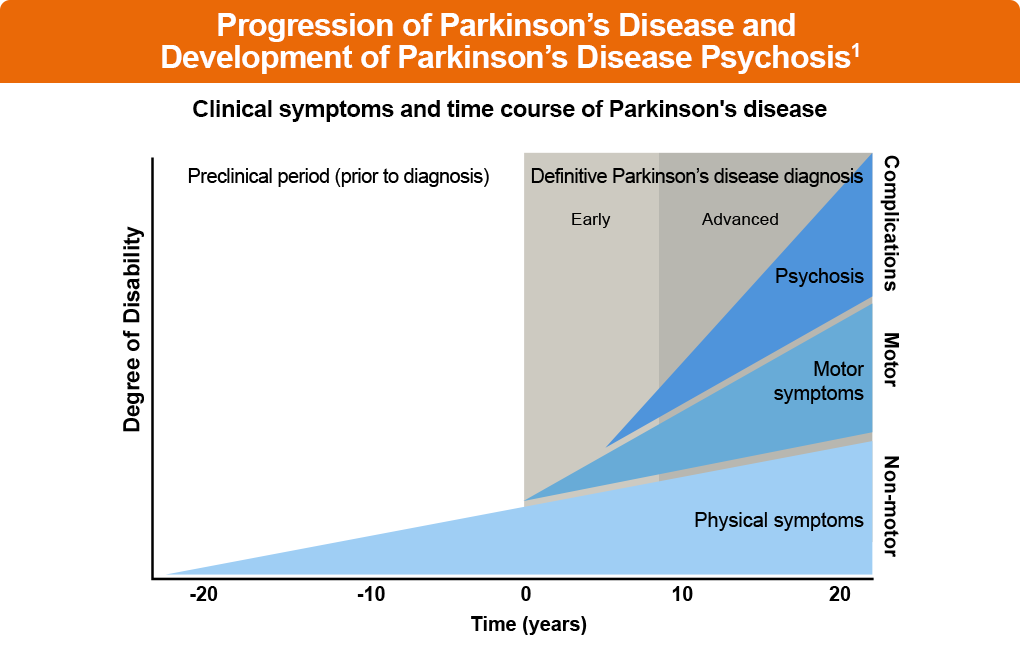
The THRIVE Patient Toolkit is a resource center for patients who received diagnosis of or who are interested in learning about Parkinson’s disease. Choose from the options below to learn more.
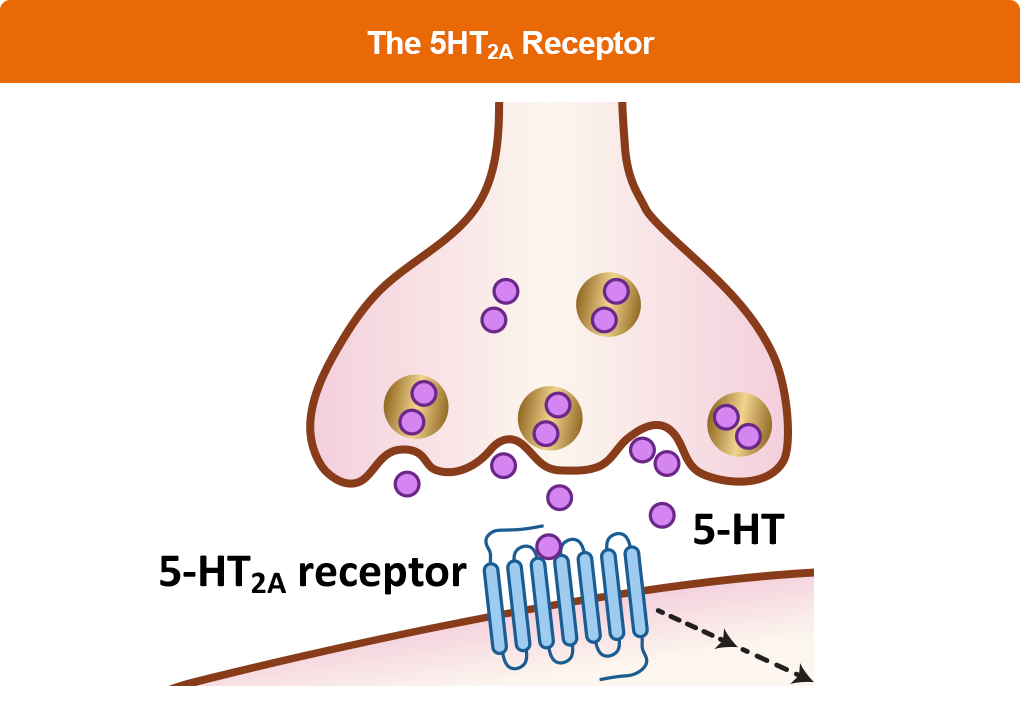
The THRIVE Clinical Toolkit is an online tool that aims to provide clinicians up-to-date information on the presentation, prognosis, pathophysiology, and treatment strategies for Parkinson’s disease. Click on one of the options below to learn more about PD.
The THRIVE Patient Toolkit is a resource center for patients who received diagnosis of or who are interested in learning about Parkinson’s disease. Choose from the options below to learn more.
The THRIVE Clinical Toolkit is an online tool that aims to provide clinicians up-to-date information on the presentation, prognosis, pathophysiology, and treatment strategies for Parkinson’s disease. Click on one of the options below to learn more about PD.
Clinical Toolkit
Patient Toolkit
Clinical Toolkit
Patient Toolkit
This activity is provided by Med Learning Group. This activity is co-provided by Ultimate Medical Academy/CCM.
This activity is supported by independent educational grants from AbbVie Inc. and Sunovion.

Copyright © 2019 Thrive Parkinsons. Built by Divigner. All Rights Reserved.
Copyright © 2019 Thrive Parkinsons. Built by Divigner. All Rights Reserved.